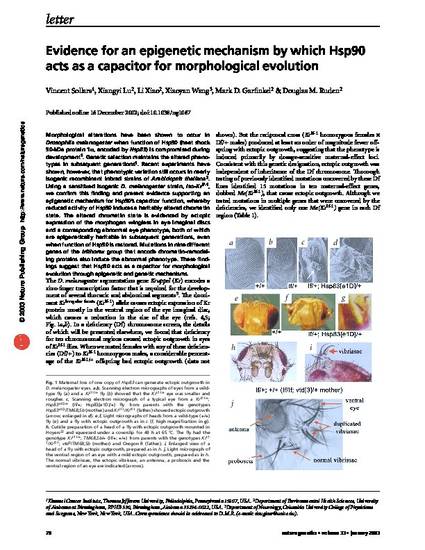
Morphological alterations have been shown to occur in Drosophila melanogaster when function of Hsp90 (heat shock 0-kDa protein 1α, encoded by Hsp83) is compromised during development1. Genetic selection maintains the altered phenotypes in subsequent generations1. Recent experiments have shown, however, that phenotypic variation still occurs in nearly isogenic recombinant inbred strains of Arabidopsis thaliana2. Using a sensitized isogenic D. melanogaster strain, iso-KrIf-1, we confirm this finding and present evidence supporting an epigenetic mechanism for Hsp90’s capacitor function, whereby reduced activity of Hsp90 induces a heritably altered chromatin state. The altered chromatin state is evidenced by ectopic expression of the morphogen wingless in eye imaginal discs and a corresponding abnormal eye phenotype, both of which are epigenetically heritable in subsequent generations, even when function of Hsp90 is restored. Mutations in nine different genes of the trithorax group that encode chromatin-remodeling proteins also induce the abnormal phenotype. These findings suggest that Hsp90 acts as a capacitor for morphological evolution through epigenetic and genetic mechanisms.

Evidence for an epigenetic mechanism by which Hsp90 acts as a capacitor for morphological evolution (2003). Vincent Sollars, Xiangyi Lu, Li Xiao, Xiaoyan Wang, Mark D. Garfinkel, and Douglas M. Ruden. Nature Genetics 33, pp. 70-74.
doi:10.1038/ng1067
This article is available on the Open Web from Nature Publishing Group and it is also included in ScienceDirect Free and Delayed Access Titles. All Rights Reserved.