Article
Multitarget 1,4-Dioxane Compounds Combining Favorable D2-like and 5-HT1A Receptor Interactions with Potential for the Treatment of Parkinson’s Disease or Schizophrenia
ACS Chemical Neuroscience
(2019)
Abstract
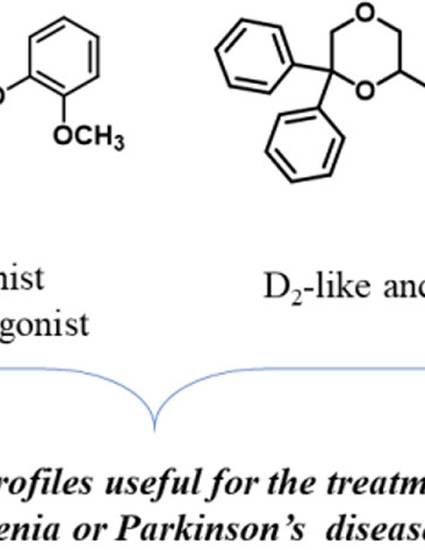
The effect of methoxy and hydroxy substitutions in different positions of the phenoxy moiety of the N-((6,6-diphenyl-1,4-dioxan-2-yl)methyl)-2-phenoxyethan-1-amine scaffold on the affinity/activity for D2-like, 5-HT1A, and α1-adrenoceptor subtypes was evaluated. Multitarget compounds with suitable combinations of dopaminergic and serotoninergic profiles were discovered. In particular, the 2-methoxy derivative 3 showed a multitarget combination of 5-HT1A/D4 agonism and D2/D3/5-HT2A antagonism, which may be a favorable profile for the treatment of schizophrenia. Interestingly, the 3-hydroxy derivative 8 behaved as a partial agonist at D2 and as a potent full agonist at D3 and D4 subtypes. In addition to its potent 5-HT1A receptor agonism, such a dopaminergic profile makes 8 a potential multitarget compound for the treatment of Parkinson’s disease (PD). Indeed, the activation of 5-HT1A receptors might be helpful in reducing dyskinetic side effects associated with dopaminergic stimulation.
Disciplines
Publication Date
May 15, 2019
DOI
10.1021/acschemneuro.8b00677
Citation Information
Fabio Del Bello, Dario Ambrosini, Alessandro Bonifazi, Amy Hauck Newman, et al.. "Multitarget 1,4-Dioxane Compounds Combining Favorable D2-like and 5-HT1A Receptor Interactions with Potential for the Treatment of Parkinson’s Disease or Schizophrenia" ACS Chemical Neuroscience Vol. 10 Iss. 5 (2019) p. 2222 - 2228 Available at: http://works.bepress.com/thomas-keck/15/
