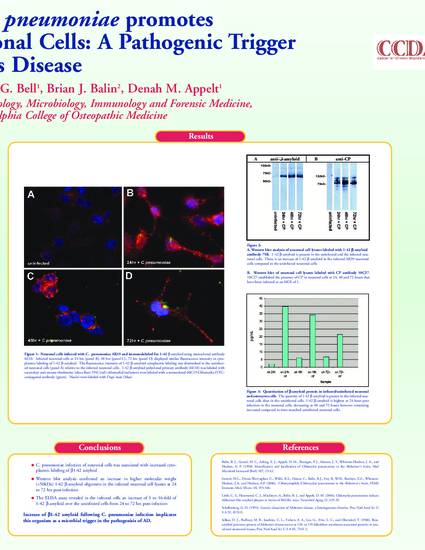
Background: Previously, our laboratory identified Chlamydophila (Chlamydia) pneumoniae (Cpn) in autopsied sporadic AD brains. Furthermore, we have developed a BALB/c mouse model that demonstrated infection-induced amyloid plaques similar to those found in AD, and demonstrated that Cpn infection of neuronal cells inhibited apoptotic pathways of cell death. Hypothesis: Our current studies address whether infection with Cpn in neuronal cells triggers abnormal cleavage of the beta amyloid precursor protein (bAPP) into Ab1-42, thereby contributing to amyloid plaque formation characteristic of the pathology identified in AD. Materials and Methods: Human neuroblastoma cells were infected with the respiratory strain AR39 Cpn in vitro, then amyloid processing was analyzed and quantitated using immunocytochemistry, Western blotting and ELISA assays. Results: Cpn was shown to infect neuronal cells and induce intracellular amyloid processing. Cpn infection yielded cytoplasmic labeling of Ab 1-42 that was increased relative to uninfected cells. The ELISA assay revealed that in neuronal cell lysates, Ab 1-42 in the infected cells was increased 3 to 16-fold over the uninfected cells, from 24 to 72hr post infection. Western blot analysis confirmed an increase in Ab 1-42 in the infected neuronal cell lysates. Conclusions: These data suggest that infection of neuronal cells with Chlamydophila (Chlamydia) pneumoniae alters the processing of bAPP, thereby producing Ab1-42. Therefore, these studies and previous research reported by our laboratory support the implication of Cpn as a pathogenic agent in perpetuating the hallmark amyloid plaque formations observed in AD. This concept holds major therapeutic considerations for future studies.
Available at: http://works.bepress.com/brian_balin/22/
